VETERINARSKI ARHIV 68 (3), 101-107, 1998
ISSN 0372-5480
Printed in Croatia
Histoplasma farciminosum infection of horses in Iraq
Falah K. Al-Ani1*, Ali H. Ali2, and Hawa B. Banna3
1Department of Veterinary Clinical Sciences, Faculty of Veterinary Medicine,
Jordan
University of Science and Technology, Irbid, Jordan
2Department of Medicine and Therapeutics, College of Veterinary Medicine, Baghdad University, Iraq.
3Department of Anatomy, College of Medicine, Baghdad University, Iraq
* Contact address:
Prof. Dr. Falah K. Al-Ani,
Department of Veterinary Clinical Sciences, Faculty of Veterinary Medicine, Jordan University of Science and Technology, Irbid, P.O. Box: 620026 Southdistrict, Jordan,
Phone: 962 2 295-111, Fax: 962 2 295-123, E-mail: alani@just.edu.jo
Al-Ani, F. K., A. H. Ali, H. B. Banna: Histoplasma farciminosum infection of horses in Iraq. Vet. arhiv 68, 101-107, 1998.
ABSTRACT
Twelve cases of naturally occurring epizootic lymphangitis with cutaneous lesions were presented. Samples of pus were taken from unopened nodules for culture and microscopy. Cutaneous tissue was submitted for histopathology and electron microscopic studies. Culture materials and direct smear stain revealed gram-positive oval or pear-shaped, yeast-like cells. Histologically, there were severe inflammatory reactions, oedema and necrosis of the examined tissue. H. farciminosum cells appeared as oval bodies. The detailed structure of the fungus was studied by electron microscopy.
Key words: Histoplasma farciminosum, epizootic lymphangitis, pathology, Iraq
Introduction
Both Histoplasma capsulatum and H. farciminosum cause clinical disease in horses (Radostits et al., 1994). Histoplasmosis caused by H. capsulatum has been reported in horses in the United States (Jungerman and Schwartzman, 1972). The infection rate varies with the geographic area and the age of the animal. On the other hand, there have also been several reports which indicate that epizootic lymphangitis is present worldwide, with foci in North Africa and Asia occurring in endemic form (Guerin et al., 1992; Jabal et al., 1983; Jerabek, 1994; Singh and Varmant, 1966). In Iraq, a study involving 1,858 Arabian-breed horses reported an epizootic lymphangitis incidence of 6.4% (Al-Ani and Al-Delaimi, 1986). However, a number of other cases remain asymptomatic. The disease is characterised by cording of the subcutaneous lymphatic, especially of limbs and neck, and the development of a series of granulating abscesses, the discharge from which is, in the form of spherical, ovoid or pear-shaped bodies, Histoplasma farciminosum detected. Light microscopy shows oval-shaped bodies of 2-3 µm in diameter. They occur extra-cellularly and intra-cellularly in macrophages (Jubb et al., 1985). Multi-nucleated cells loaded with the organisms have also been recorded (Jones and Hunt, 1983; Singh, 1956). The purpose of this paper is to report the clinical signs and pathological changes in horses with epizootic lymphangitis, and to present morphological details of H. farciminosum.
Materials and methodsHorses
Twelve horses with cutaneous lesions of epizootic lymphangitis were presented to the Large Animal Clinic, College of Veterinary Medicine, Baghdad University. All were Arabian-breed horses. They were kept in a large-scale racehorse maintenance facility. A routine clinical examination was carried out. Skin, eyes and nasal passages were carefully examined for any lesions. Samples for direct smear staining, culture and pathological studies were submitted.
Microbiological examination
Closed nodular lesions were first cleaned with ethanol and the purulent content was aspirated with a sterile syringe. Fresh smears were prepared and stained with Gram-stain and Giemsa's stain. Cultures were made from the purulent material on mycobiotic agar (Difco) and incubated at 26 oC. The growth was observed for 42 days. The media were checked every 2-3 days and microscope slides were prepared from growing colonies in lactophenol cotton blue. For bacteriological isolation, pus samples were streaked on blood agar plates and incubated at 37 oC.
Pathological study
For the collecting of samples for pathological study, the horses were lightly sedated and tissue samples were taken from cutaneous lesions using a sharp surgical blade. The tissue samples were divided into two parts. One part was fixed in 10% buffered neutral formalin and paraffin sections, 6 µm thick, were prepared by standard methods and stained by haematoxylin and eosin (H&E) stain. The other skin biopsy sample was cut into small pieces (1.5-2.0 mm) with a sharp blade and immediately prefixed in phosphate buffered 2% glutaraldehyde solution at 4 oC and postfixes in 1% osmium tetroxide (OsO4). Ultra-thin sections were cut using glass knives by "Ultracut Reichert" (Vienna, Austria). The sections were then double stained with uranyl acetate and lead citrate and examined by a Philips-300 electron microscope at 60 kV and photographed.
Results
Clinical signs
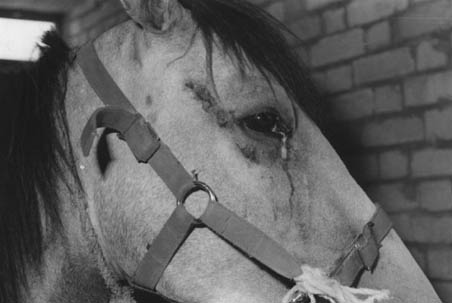
Two different clinical forms of epizootic lymphangitis were observed in the 12 horses in this study. The cutaneous form was seen in eleven horses. Cutaneous lesions were often limited to certain areas of the body, such as the face, the anterior aspect of the front legs, the chest wall and the neck (Fig. 1). The lesions began to develop indolent chancre-like papules, and over the course of some weeks became larger and irregular. The superficial lymphatic was thickened and all ages of abscessing were observed along the course of lymph vessels. In one case, a lesion developed on the conjunctiva and the nictitating membrane, initially producing a small papule giving off a serous ocular discharge, followed by severe ulcerative keratoconjunctivitis.

Fig. 1. Cutaneous lesions on the face of a horse suffering of epizootic lymphangitis
Microbiological isolation
A yellowish-green coloured pus was collected from the nodules. Direct smear stain revealed gram-positive oval or pear-shaped yeast-like cells (Fig. 2). In mycobiotic agar at 26 oC, the growth of primary isolates was evident after 2-3 weeks of incubation. The colonies appeared as being minute, circular, raised and greyish in colour, with a smooth, glistening surface. After further incubation the colonies turned light with raised, rough edges and lighter in colour at the periphery (Fig. 3). Smears prepared from these colonies and stained with lactophenol stain showed the presence of the typical yeast form H. farciminosum. No bacterial isolation was obtained.

Fig. 2. Gram-stain smear of Histoplasma farciminosum from the nodule
of
a horse with epizootic lymphangitis
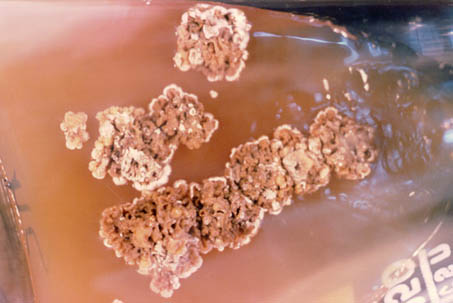
Fig. 3. Raised, rough edges, lighter in colour at periphery, adherent colonies of H. farciminosum in mycobiotic agar after 42 days incubation at 26 °C
Pathological studies
Severe inflammatory reaction, oedema and necrosis were observed in all samples examined. Some nodules had predominantly polymorphonuclear leukocytes in the subcutaneous tissues. In the greater majority of nodules, the most prominent infiltrated cells were mononuclear. Under light microscope, H. farciminosum appeared usually as oval bodies ranging from 2-4 µm (Fig. 4). Most of the details of the fine internal structure observed by electron microscopy involved the cell envelope, plasma membrane, cell wall, capsule and inner cell structures (Fig. 5). The cell envelope (CE) comprised the plasma membrane, cell wall, and capsule. The plasma membrane (PM) was the outer limit of the cytoplasm. The cell wall (CW) was external to the plasma membrane. The nucleus (N) was located peripherally at a lobe that was irregular in shape and varied in size.
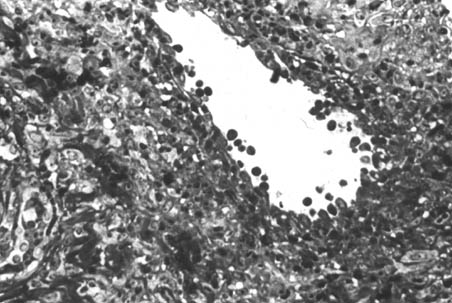
Fig. 4. Histopathological section through the cutaneous nodule
from a horse
with epizootic lymphangitis (H&E stain)
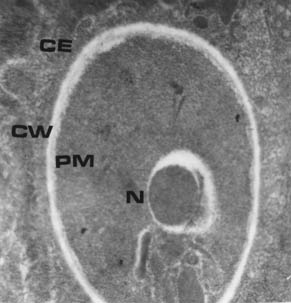
Fig. 5. Electron micrograph of H. farciminosum showing the cell envelope (CE), plasma membrane (PM), cells wall (CW) and the nucleus (N). × 25,000.
Discussion
Epizootic lymphangitis is present worldwide, with foci in North Africa and Asia and occurring in endemic forms (Singh, 1956; Fawi, 1969). The clinical presentation of epizootic lymphangitis ranged from animals that were asymptomatic carrier, to animals with widely disseminated skin lesions (Al-Ani and Al-Delaimi, 1986). The cutaneous form is that most commonly seen. However, there are reports of the ocular and respiratory forms (Fawi, 1969; Khater et al., 1968).
The principal test used for diagnosis of epizootic lymphangitis is stained smears of the cutaneous exudate (Al-Ani and Al-Delaimi, 1986). Sections of cutaneous lesions revealed granulomatous reactions similar to those reported previously (Jones and Hunt, 1983; Jubb et al., 1985). H. farciminosum was found to possess ultra-structural characteristics similar to those reported in Cryptococcus neoformans (Edwards et al., 1967). The main differences being that H. farciminosum had thick cell walls and that the nuclear material was not obvious.
The lesions, which were localised in the skin, revealed granulomatous tissue reactions with a predominance of large macrophages. However, some nodules had poly-morphonuclear cells. The causative organisms were principally located within histocytes, appearing as oval organism of 2×3 µm and enveloped by a thin capsule (Jones and Hunt, 1983). Although the fungus can be demonstrated by H&E stain in the tissue sections, special stains may be of value. Staining by PAS or Gridley's fungus stain identifies the capsule selectively, staining it red and leaving the central body unstained (Jones and Hunt, 1983).
References
Al-Ani, F. K., A. K. Al-Delaimi (1986): Epizootic lymphangitis in horses: clinical, epidemiological and hematological studies. Pakistan Vet. J. 6, 96-99.
Edwards, M. R., M. A. Gorden, E. W. Lapa, W. C. Ghiorss (1967): Micromorphology of Cryptococcus neoformans. J. Bacteriol. 94, 766-777.
Fawi, M. T. (1969): Fluorescent antibody test for the serodiagnosis of Histoplasma farciminosum infection in equidae. Brit. Vet. J. 125, 231-234.
Guerin, C., S. Abebe, F. Touati (1992): Epizootic lymphangitis in horses in Ethiopia. Med. Mycology. 2, 1-5.
Jabal, M. A., F. K. Hassan, A. A. Siad, K. A. Karim (1983): Studies of equine Histoplasmosis farciminosum and characterization of Histoplasma farciminosum. Sabouraudia 21, 124-127.
Jerabek, J. (1994): Less known animal diseases: epizootic lymphangitis. Veterinarstvi 44, 348-350.
Jones, T. C., R. D. Hunt (1983): Veterinary Pathology, 5th ed. Lea & Febiger. Philadelphia.
Jubb, K. V., P. C. Kennedy, N. Palmer (1985): Pathology of Domestic Animals, Volume 3, 3rd ed. Academics press. New York.
Jungerman, P. F., R. M. Schwartzman (1972): Veterinary Medical Mycology. Lea & Febiger. Philadelphia.
Khater, A. R., M. Iskander, A. Mostafa (1968): A histomorphological study of cutaneous lesions in equine histoplasmosis (epizootic lymphangitis). J. Egypt Vet. Med. Ass. 28, 165-174.
Radostits, O. M., D. C. Blood, C. C. Gay (1994): Veterinary Medicine, 8th ed. Bailliere Tindall. London.
Singh, M. S. (1956): Equine cryptoccosis (epizootic lymphangitis). Indian Vet J. 32, 260-267.
Singh, T. E., B. M. Varmant (1966): Studies on epizootic lymphangitis. A note on pathogenicity of Histoplasma farciminosum for laboratory animals. Indian J. Vet. Anim. Husb. 36, 164-167.
Received: 23 February 1998
Accepted: 25 April 1998
Al-Ani, F. K., A. H. Ali, H. B. Banna: Infekcija konja gljivicom Histoplasma farciminosum u Iraku. Vet. arhiv 68, 101-107, 1998.
SAZETAK
Prikazana je pojava epizootskog limfangitisa s promjenama na kozi u 12 prirodno oboljelih konja. Uzorci gnoja za mikroskopsku i kulturelnu pretragu bili su uzeti iz neotvorenih cvorica. Kozno tkivo bilo je pretrazeno histopatoloski i elektronskomikroskopski. U razmascima s kulture i u izravno obojenim razmascima iz materijala dokazane su gram-pozitivne, ovalne ili kruskolike stanice slicne kvascima. Histoloski je ustanovljena jaka upalna reakcija, edem i nekroza pretrazenog tkiva. Stanice H. farciminosum izgledale su kao ovalna tjelesca. Detaljna grada ove gljivice proucavana je elektronskomikroskopski.
Kljucne rijeci: Histoplasma farciminosum, epizootski limfangitis, patologija, Irak